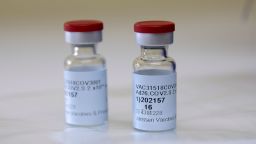
A third vaccine is poised to join the fight against Covid-19, with 3.9 million doses expected to be available right away, a health official said Friday.
That amount could increase vaccinations for states by 25% and would be delivered in as little as one or two days in the first week, said Lori Tremmel Freeman, the chief executive officer of the National Association of County and City Health Officials.
The US Food and Drug Administration on Saturday authorized Johnson & Johnson’s single-dose vaccine for emergency use in adults 18 and older, meaning the vaccine may be available early next week.
An FDA advisory committee Friday recommended the authorization.
Vaccine administration already has been increasing, with 2.2 million more vaccinations reported Friday than the day before and about 70.5 million doses given in total, according to the US Centers for Disease Control and Prevention.
But with variants spreading and threatening to send new case rates skyrocketing once more, officials hope to get ahead of the spread with faster inoculations.
“We’ve had two vaccines, and now it looks like we are going to get three. And that means we can get more doses into arms and we can try to get this terrible pandemic behind us,” Dr. Francis Collins, director of the National Institutes of Health, told CNN’s Wolf Blitzer.
New coronavirus cases have begun to plateau after a steady decline. CDC Director Dr. Rochelle Walensky warned it could be the “beginning effects” of more transmissible variants having an impact.
“CDC has been sounding the alarm about the continued spread of variants in the United States,” she said during a White House briefing on Friday.
Misconceptions that Johnson & Johnson vaccine is ‘second class’
The vaccine, made by Janssen, J&J’s vaccine arm, doesn’t require special storage.
The 22 members of the FDA’s Vaccines and Related Biological Products Advisory Committee voted unanimously to recommend the Johnson & Johnson vaccine, which one member said was an “easy call.”
“It clearly gets way over the bar and it’s nice to have a single-dose vaccine,” said Dr. Eric Rubin, editor-in-chief of the New England Journal of Medicine and a professor at Harvard University’s T.H. Chan School of Public Health.
But some officials worry that the public views it as “second class,” a misconception public health leaders will need to address.
“It’s difficult to make an apples-to-apples comparison between vaccines authorized, based on data collected before new variants are believed to have been in widespread circulation,” said Sarah Christopherson, the policy advocacy director at the National Women’s Health Network.
Johnson & Johnson’s vaccine may appear to have a lower efficacy rate than its earlier counterparts. But that does not make it a worse option because it appears to protect against some variants, another member of the committee to recommend the vaccine said.
“One dose will keep you out of the hospital, keep you out of the intensive care unit and keep you out of the morgue,” Dr. Paul Offit told Blitzer.
Several public health experts told Congress on Friday that people who have the Johnson & Johnson vaccine made available to them should get it.
“If I had a J&J vaccine available today and a Moderna vaccine available tomorrow, I would be happy to take the J&J today. I don’t feel like I would need to wait. They are all terrific vaccines for the things that we care about,” Dr. Ashish Jha, dean of the Brown University School of Public Health, told a House Ways and Means health subcommittee hearing.
Not the time to change doses
Promising news also has come out for the Pfizer/BioNTech vaccine.
Just one dose can induce a strong enough immune response in people who have already had Covid-19 that it could protect from future infection, according to two papers published in the journal The Lancet on Thursday.
The Pfizer vaccine is currently administered as two doses 21 days apart. The first dose primes the immune system and the second boosts it.
Some officials have suggested prioritizing the administration of first doses to increase the immune response in as many people as possible quickly.
But with emerging variants, now is not the time to change the two-dose schedule for Pfizer and Moderna vaccines, said Dr. Nancy Messonnier, the director of the National Center for Immunization and Respiratory Diseases at the CDC, in a conversation with the Journal of the American Medical Association on Friday.
“The vaccines have been studied and approved, authorized, recommended as a two-dose schedule. Our programs are built on that. We’ve communicated that to the public,” she said. “I just don’t think that there is enough science yet to tell us that it’s a moment to change what we know to be an effective regimen.”
CNN’s Jen Christensen, Maggie Fox, Jamie Gumbrecht, Jacqueline Howard, Virginia Langmaid, Lauren Mascarenhas, Deidre McPhillips and Nicholas Neville contributed to this report.